Internal Routing
The Office of Research and Sponsored Programs (ORSP) offers services that are designed to assist faculty in proposal development and to facilitate the overall grant process, while balancing the need for institutional and sponsor regulatory compliance. This page provides a listing of resources to guide investigators as they seek internal approvals before proposal submission.
Internal Routing & Approval
All proposals for external funding must be routed and approved by the University administration, regardless if UL Lafayette is the Prime Applicant or Subrecipient. Submission of a proposal without all required documentation will delay processing of that proposal and may result in the proposal not being submitted to the funding agency. ORSP cannot guarantee processing and submission of a proposal if a complete proposal package is received by ORSP after the internal deadline.
For standard proposals, the 10-5-3 timeline gives ORSP adequate time to assist you in submitting a proposal with the least amount of errors. It’s designed to reduce stress for PIs, enhance collaboration with ORSP staff, and ensure proposals meet institutional, state, and agency requirements.
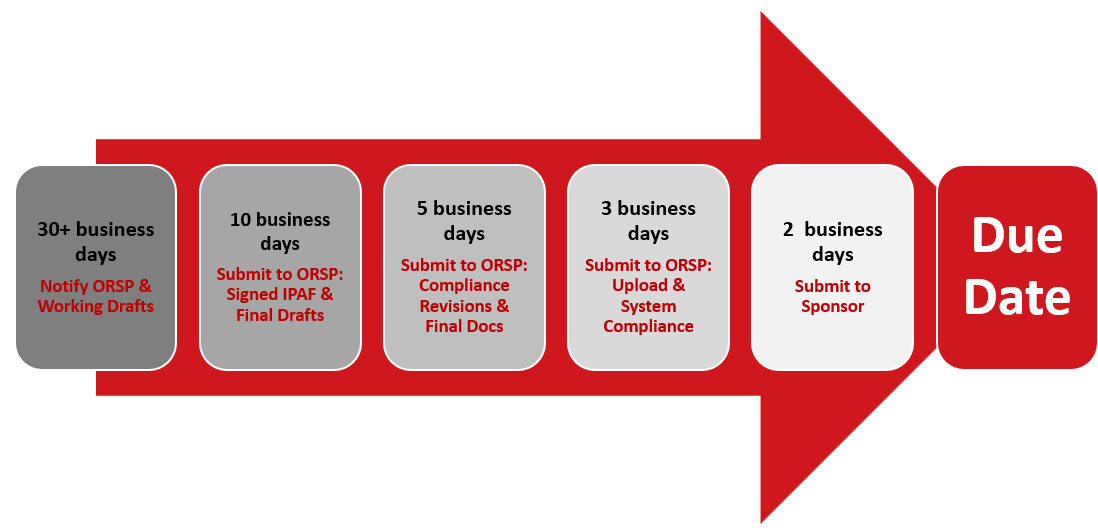
|
30+ Business Days Prior to agency deadline |
|
|
10 Business Days Prior to agency deadline |
|
|
5 Business Days Prior to agency deadline |
|
|
3 Business Days Prior to agency deadline |
|
|
2 Business Days Prior to agency deadline |
|
Proposal Categories: Standard, Complex, and Limited Submissions
• Standard Proposals: No subawards and total budget under $1 million.
• Complex Proposals: Include subawards, exceed $1 million, or deviate from standard agency guidelines.
• Limited Submission Proposals: Agency restricts the number of submissions per institution; an internal selection process is required.
Customized timelines will be provided for complex and limited submission proposals. Please consult with your Pre-Award Grants Specialist early in the planning process.
A full proposal package includes all documentation required by the agency for submission plus all internal approval forms. ORSP has compiled a comprehensive checklist (both electronic and printer-friendly) for for the components that comprise a complete proposal package. For listing details, please refer to the links below:
Internal Routing Forms
Below is a listing of our downloadable forms and a description of each form. If you have any questions about whether a form is required or how to fill out the form, please contact your Pre-Award Administrator.
|
Form |
Description |
|
Also commonly referred to as a “routing form,” this form is required of all proposal submissions and must be sent with the narrative, budget, and budget justification (at a minimum) for your proposal. This form must be completed and signed by all investigators and their respective Department Heads, Deans, and/or Center Directors. Please note: This form is an internal document and will not be submitted to funding agencies or collaborators. ORSP has created an instructions document to help you fill out the IPAF or routing form. |
|
|
Use the Additional Investigator Attachment Form should you have more investigators than the Internal Routing Form can accommodate. Please note that all investigators and their respective Department Heads, Deans, and/or Center Directors must approve the proposal before it is routed to ORSP. |
|
|
The submittal of the F&A Cost Waiver/Reduction Request Form is required only on those rare occasions in which the PI requests a waiver or reduction of the F&A rate below the maximum allowed by the sponsor. This form is NOT required if the F&A rate in the proposal is the maximum permitted by the sponsor’s written policy, even when that rate is lower than the University’s federally negotiated/approved rate. In those cases, attach a copy of the sponsor’s policy to the proposal for routing. |
|
|
The Cost Share/Matching Funds Approval Form is required when the proposal budget includes cost sharing or matching funds. |
|
|
Required for all investigators for NSF & PHS (including NIH) proposals or for an individual investigator if a financial conflict of interest exists, regardless of sponsor. |
